Search
To determine the efficacy of advanced hybrid closed-loop therapy in a high-risk cohort of youth on continuous subcutaneous insulin infusion with or without continuous glucose monitoring with suboptimal glycemia.
To explore parents' experiences of using continuous glucose monitoring in their young children with early-stage type 1 diabetes, being followed in the Australian Environmental Determinants of Islet Autoimmunity (ENDIA) study.
Dietary patterns characterised by high intakes of vegetables may lower the risk of pre-eclampsia and premature birth in the general population. The effect of dietary patterns in women with type 1 diabetes, who have an increased risk of complications in pregnancy, is not known.
The evidence about the acceptability and effectiveness of innovative paediatric models of care for Type 1 diabetes is limited. To address this gap, we synthesised literature on implemented models of care, model components, outcomes, and determinants of implementation and sustainability.
One third of Australian children diagnosed with type 1 diabetes present with life-threatening diabetic ketoacidosis (DKA) at diagnosis. Screening for early-stage, presymptomatic type 1 diabetes, with ongoing follow-up, can substantially reduce this risk (<5% risk). Several screening models are being trialled internationally, without consensus on the optimal approach. This pilot study aims to assess three models for a routine, population-wide screening programme in Australia.
The ESCALATION system is a novel paediatric Early Warning System that incorporates family involvement and sepsis recognition. This study aimed to assess the feasibility and iteratively refine the ESCALATION system in a variety of hospital settings in preparation for full-service implementation.
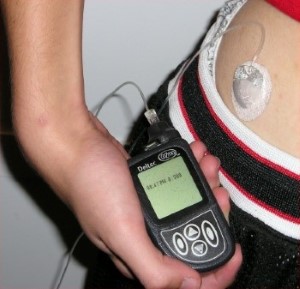
What is active insulin and why is it so important to diabetes management?

An insulin pump can halve children’s exposure to hypoglycaemia if it includes an automated system to suspend basal insulin when hypoglycaemia is predicted
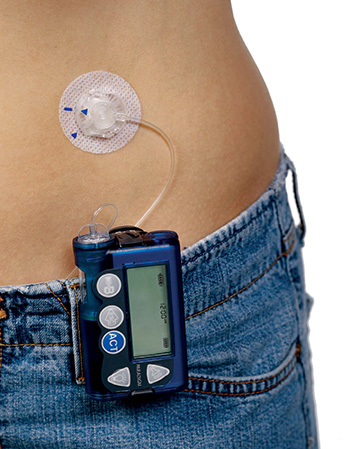
What to do in the event of a pump failure.

Read on to find out how to link the Dexcom G5 to a Diasend account.
