Search

New research from the Children’s Diabetes Centre has found a pump that suspends insulin delivery when BGLs fall can halve the incidence of hypoglycaemia.
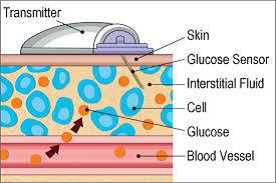
The Children’s Diabetes Centre is now recruiting for a new continuous glucose monitoring study.

Parents of children with Type 1 Diabetes often feel they need to wrap their child in cotton wool rather than let them get on with the job of being a kid and eve

Participants are needed for a new study looking at whether protein after exercise can decrease the risk of delayed low blood glucose levels after exercise.

April Welsh lives life in top gear, literally. Diagnosed with T1D just before her 4th birthday, April became the first female to race in the Formula Vee

For five years, Dr Martin de Bock has called Perth home. Now, it is time for him to return to his birthplace — the green pastures of Auckland, New Zealand.

A record Perth crowd turned out for JDRF’s One Walk to raise much-needed funds for Type 1 Diabetes (T1D) research last month.

The school holidays and Christmas are a welcome break for most families but the lack of routine can prove a trying time for families living with Type 1 Diabetes

Doctors are worried by a record number of WA children diagnosed with type 1 diabetes this year, with about 30 per cent more cases than average.

Evidence is mounting that the bacteria in a mother’s digestive system, known as microbiome, can influence the health of babies.
