Search
Showing results for "clinical trials"
Within PF-EPN-A, 1q gain is a marker of poor prognosis, however, it is unclear if within PF-EPN-A additional cytogenetic events exist which can refine risk stratification.
Nick Gottardo MBChB FRACP PhD Head of Paediatric and Adolescent Oncology and Haematology, Perth Children’s Hospital; Co-head, Brain Tumour Research
The Zero Childhood Cancer Program is a precision medicine program to benefit children with poor-outcome, rare, relapsed or refractory cancer. Using tumor and germline whole genome sequencing (WGS) and RNA sequencing (RNAseq) across 252 tumors from high-risk pediatric patients with cancer, we identified 968 reportable molecular aberrations.
Medulloblastoma (MB) is the most common malignant brain tumor in children and a leading cause of cancer-related mortality and morbidity.

Head of Paediatric and Adolescent Oncology and Haematology, Perth Children’s Hospital; Co-head, Brain Tumour Research Program, The Kids Research Institute Australia
The Australasian Society of Clinical Immunology and Allergy (ASCIA) Guideline: Infant Feeding for Food Allergy Prevention is an update of the 2016 ASCIA guideline. This updated guideline provides recommendations specifically in relation to infant feeding for food allergy prevention.

A list of organisations that currently collaborate with ORIGINS.

Co-Head, Leukaemia Translational Research
SHIP-CT, led by Professor Stephen Stick, Director of the Wal-yan Respiratory Research Centre at The Kids, is a unique study in preschool-aged children (from 3-6 years of age) with CF using images of the lung from chest CT scans as the main outcome measure.
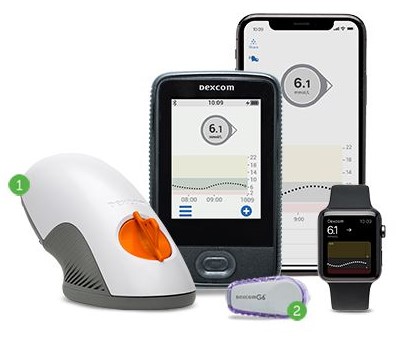
A new Q&A series focusing on the different research themes within the Children’s Diabetes Centre - technology.
