Search
Showing results for "clinical trials"
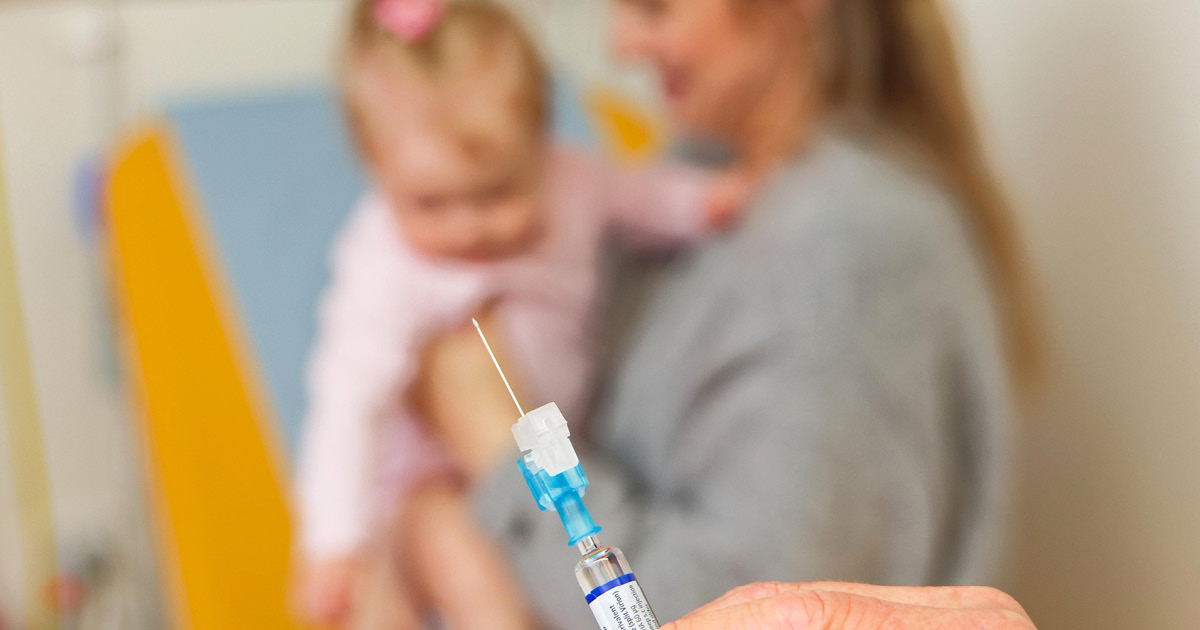
As a leading research site in Australia, the Wesfarmers Centre of Vaccines and Infectious Diseases played an instrumental role in the global effort to develop a world-first RSV immunisation for young babies.
During the early phases of the 2009 pandemic, subjects with influenza-like illness only had laboratory testing specific for the new A(H1N1)pdm09 virus.
Respiratory failure, multiple organ failure, shortness of breath, recovery, and mortality have been identified as critically important core outcomes by more than 9300 patients, health professionals, and the public from 111 countries in the global coronavirus disease 2019 core outcome set initiative. The aim of this project was to establish the core outcome measures for these domains for trials in coronavirus disease 2019. Three online consensus workshops were convened to establish outcome measures for the four core domains of respiratory failure, multiple organ failure, shortness of breath, and recovery.
The epidemiology and clinical manifestations of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection are different in children and adolescents compared with adults. Although coronavirus disease 2019 (COVID-19) appears to be less common in children, with milder disease overall, severe complications may occur, including paediatric inflammatory multisystem syndrome.
Itraconazole remains a first-line antifungal agent for certain fungal infections in children, including allergic bronchopulmonary aspergillosis (ABPA) and sporotrichosis, but poor attainment of therapeutic drug levels is frequently observed with available oral formulations. A formulation of 'SUper BioAvailability itraconazole' (SUBA-itraconazole; Lozanoc®) has been developed, with adult studies demonstrating rapid and reliable attainment of therapeutic levels, yet paediatric data are lacking.
Helen Jenny Peter Leonard Downs Jacoby MBChB MPH BApplSci (physio) MSc PhD BA (Hons) MSc Principal Research Fellow Head, Child Disability
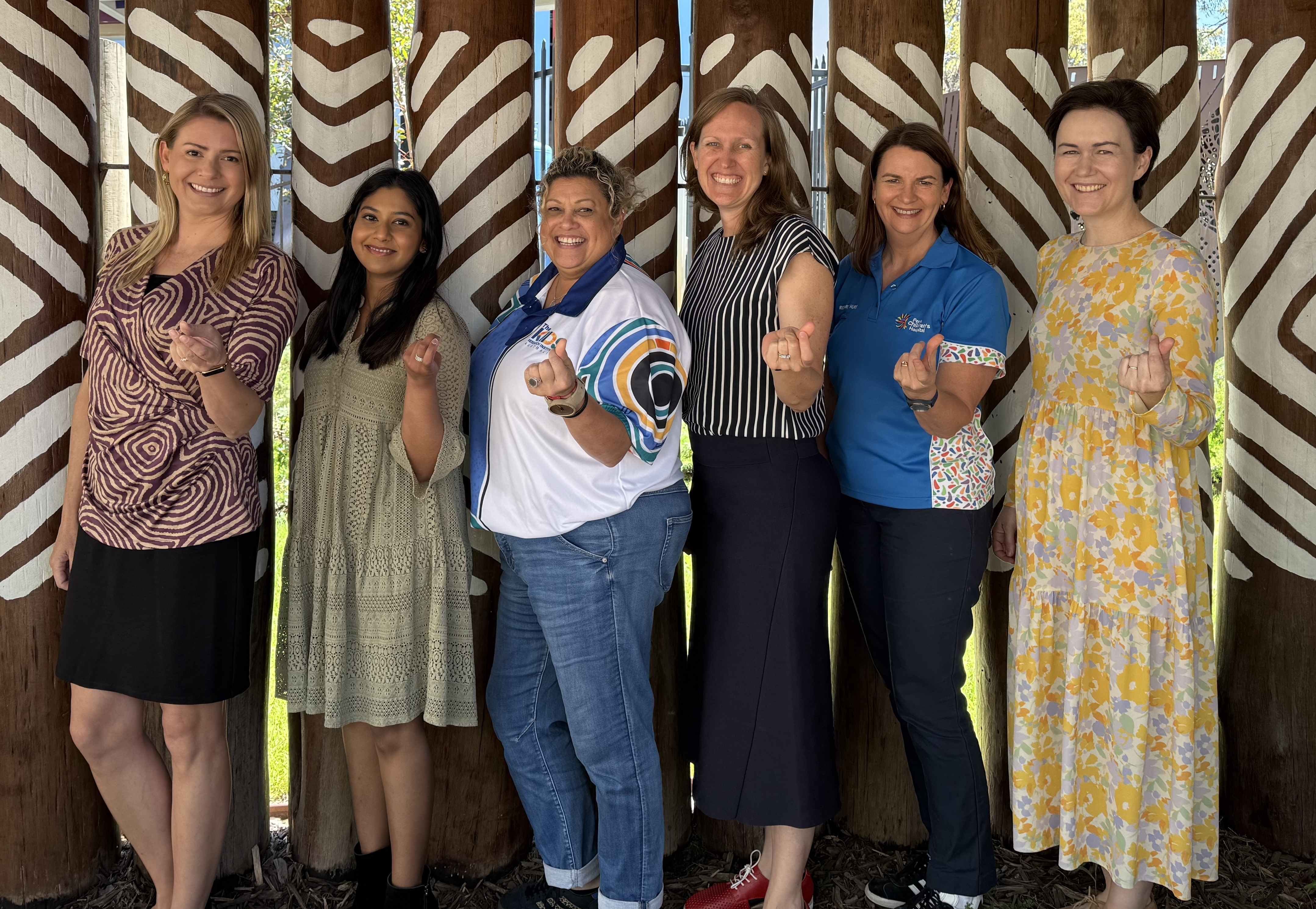
Perth investigators involved in a major global trial have launched an innovative Cultural Information Hub to maximise cultural safety for Aboriginal and/or Torres Strait Islander patients participating in research.

The Australian arm of an international clinical trial looking at improved treatments for young babies with leukaemia has been awarded funding from the MRFF.
Previous preclinical and clinical trials have shown promising antitumour activity and toxicity profile when employing the ‘Synergy between Immunotherapy and Radiotherapy’ (SITAR) strategy. Approximately, one in seven radiation therapy studies currently recruiting is investigating SITAR.
The aim of this study was to understand the challenges experienced by families obtaining a diagnosis and therapy for developmental coordination disorder (DCD). Parents of 435 children aged 4-18 years with persistent motor difficulties consistent with a diagnosis of DCD completed an online survey. Diagnostic timeline and diagnostic label/s received were examined, along with therapies accessed.
