Search
Showing results for "clinical trials"
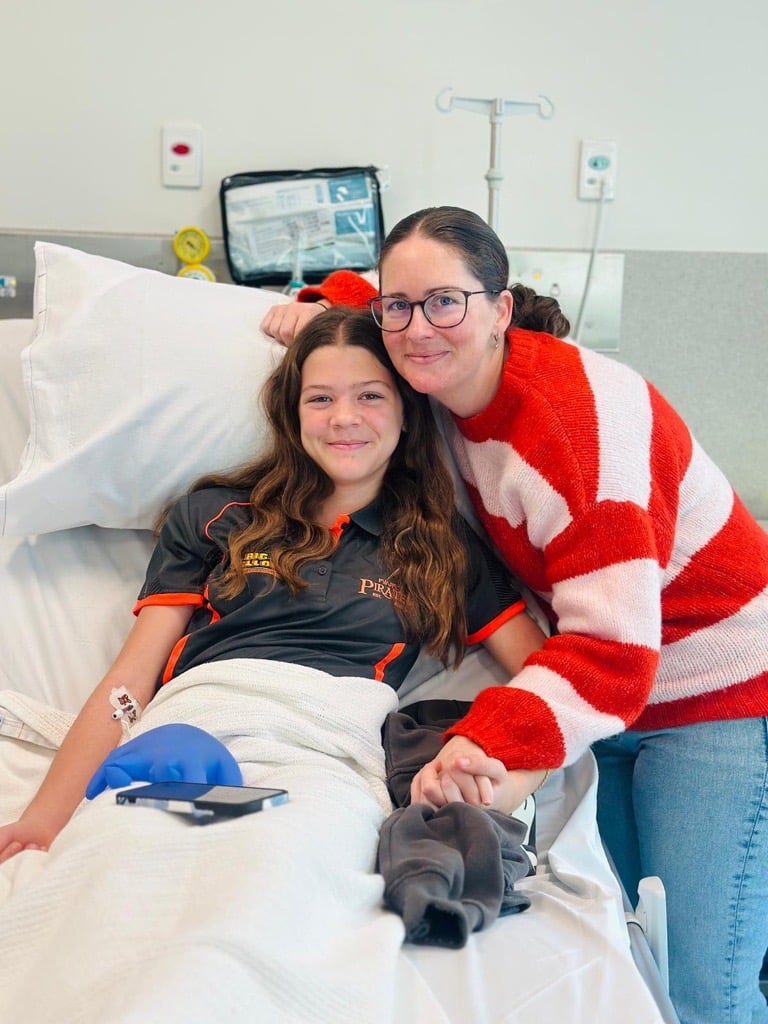
To celebrate International Clinical Trials Day, we are highlighting a couple of a current trials underway at the Children's Diabetes Centre at the Kids Research Institute Australia and Perth Children's Hospital
The Opportunity We are seeking an individual who has experience in setting up and monitoring multi-centre clinical trials for pharmaceutical
To coordinate and monitor clinical trial sites with the aim of meeting protocol and good clinical practice requirements.

Learn more about all of the Clinical Trials, Platforms & Cohorts at the Wal-yan respiratory centre.
Neonatal sepsis is a leading cause of infant mortality worldwide with non-specific and varied presentation. We aimed to catalogue the current definitions of neonatal sepsis in published randomised controlled trials.
This narrative review describes the evolution, range and relative strengths and weaknesses of endpoints used in late phase trials
Raelene Endersby BSc (Hons) PhD Brainchild Fellow; Co-Head, Brain Tumour Research raelene.endersby@thekids.org.au Co-Head, Brain Tumour Research
There is a recognized unmet need for clinical trials to provide evidence-informed care for infants, children and adolescents. This Special Communication outlines the capacity of 3 distinct trial design strategies, sequential, parallel, and a unified adult-pediatric bayesian adaptive design, to incorporate children into clinical trials and transform this current state of evidence inequity. A unified adult-pediatric whole-of-life clinical trial is demonstrated through the Staphylococcus aureus Network Adaptive Platform (SNAP) trial.
Tobias Strunk MD, PhD, FRACP Head, Neonatal Health tobias.strunk@thekids.org.au Head, Neonatal Health Clinical Professor Tobias Strunk is a
Adaptive clinical trials have designs that evolve over time because of changes to treatments or changes to the chance that participants will receive these treatments. These changes might introduce confounding that biases crude comparisons of the treatment arms and makes the results from standard reporting methods difficult to interpret for adaptive trials. To deal with this shortcoming, a reporting framework for adaptive trials was developed based on concurrently randomised cohort reporting.
